Nirsevimab immunization effectiveness against respiratory syncytial virus hospitalization in newborns: two season of use in Navarre, Spain
DOI:
https://doi.org/10.23938/ASSN.1133Keywords:
Respiratory Syncytial Virus, Prevention, Nirsevimab, Hospitalisation, EffectivenessAbstract
Background. Respiratory syncytial virus (RSV) is the leading cause of infant hospitalisation. In 2022, nirsevimab was approved in the European Union to prevent severe respiratory disease due to RSV during the first year of life. Our aim is to evaluate the effectiveness of nirsevimab immunoprophylaxis in new-borns for preventing RSV -related hospitalisations in Navarre, Spain, during its first two seasons of use.
Methods. Nirsevimab was offered free of charge to infants born from October to December 2023 and from September to December 2024. Each cohort was followed until February of the following year. Cases were infants hospitalised for PCR-confirmed RSV infection. Cox regression was used to estimate the hazard ratio of hospitalisation for immunised versus non-immunised children.
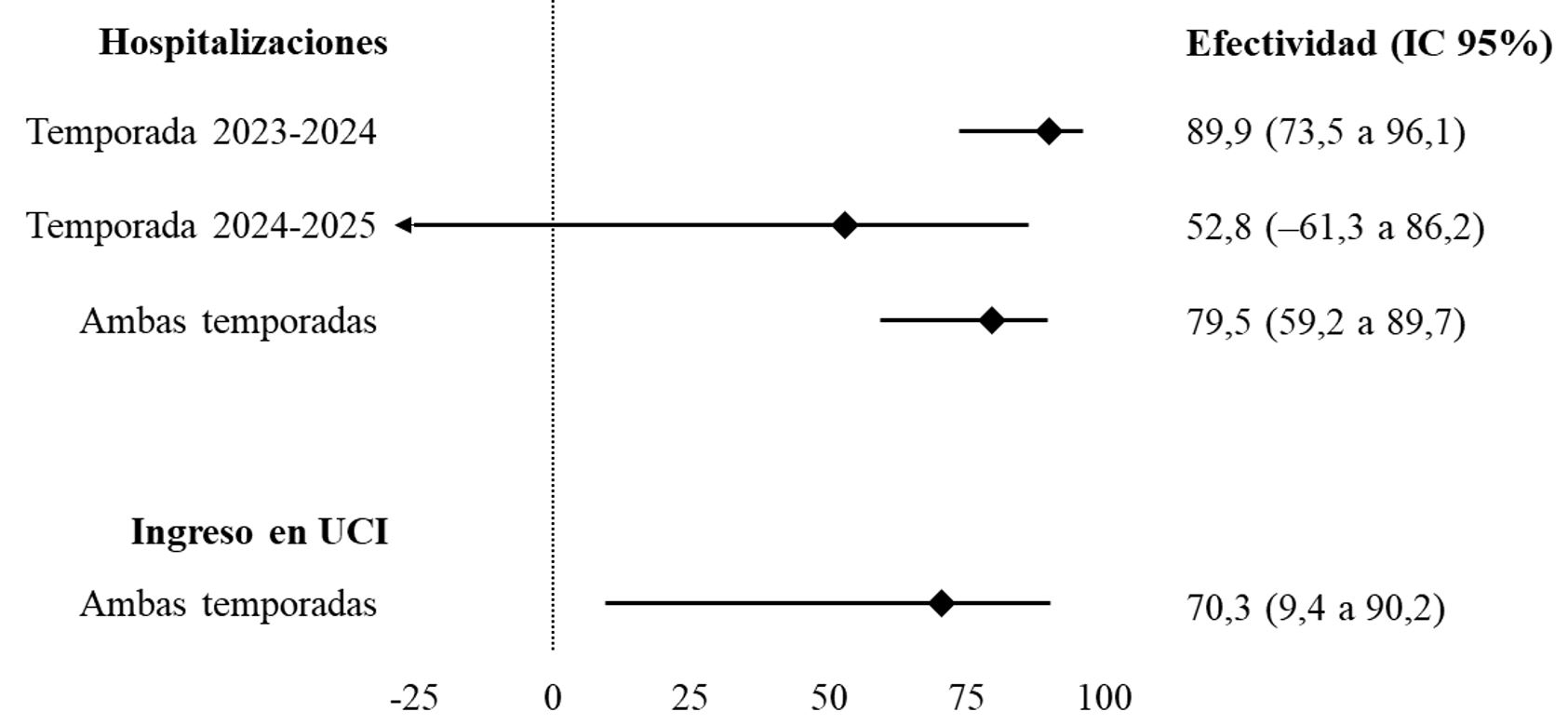
Results. Nirsevimab was offered to 2,699 new-borns; of them, 2,541 (94.1%) received nirsevimab. In the 2023-2024 season, 17 RSV-related hospitalisations were recorded and 24 in the 2024-2025 season. The average risk of RSV hospitalisation was 7.6% in non-immunised new-borns versus 1.1% in immunised ones. Overall, effectiveness of nirsevimab was 79.5% (95% CI: 59.2 - 89.7), with estimates of 89.9% in 2023-2024 and 52.8% in 2024-2025, with no significant differences between seasons (p=0.055). On average, one RSV hospitalisation was prevented per 22.6 immunised infants.
Conclusions. Nirsevimab immunoprophylaxis substantially reduces RSV hospitalisations, helping ease paediatric hospital burden. However, as some immunised infants were still hospitalised, additional preventive measures remain necessary.
Downloads
References
1. HALL CB, WEINBERG GA, IWANE MK, BLUMKIN AK, EDWARDS KM, STAAT MA et al. The burden of respiratory syncytial virus infection in young children. N Engl J Med 2009; 360(6): 588-598. https://doi.org/10.1056/NEJMoa0804877
2. LI Y, WANG X, BLAU DM, CABALLERO MT, FEIKIN DR, GILL CJ et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet 2022; 399(10340): 2047-2064. https://doi.org/10.1016/S0140-6736(22)00478-0
3. Temporada de virus respiratorios 2022-2023 en Navarra. (Septiembre de 2023). Boletín de Salud Pública de Navarra Nº 126. https://www.navarra.es/NR/rdonlyres/AECCD760-AB2A-4841-818A-FA53478FD6DC/488194/BOL126INT1.pdf
4. LANGLEY GF, ANDERSON LJ. Epidemiology and prevention of respiratory syncytial virus infections among infants and young children. Pediatr Infect Dis J 2011; 30(6): 510-517. https://doi.org/10.1097/INF.0b013e3182184ae7
5. VIGURIA N, MARTÍNEZ-BAZ I, MORENO-GALARRAGA L, SIERRASESÚMAGA L, SALCEDO B, CASTILLA J. Respiratory syncytial virus hospitalization in children in northern Spain. PLoS One 2018; 13(11): e0206474. https://doi.org/10.1371/journal.pone.0206474
6. European Medicines Agency (EMA). Beyfortus®. Consultado el 19 de febrero de 2025. https://www.ema.europa.eu/en/medicines/human/EPAR/beyfortus
7. European Medicines Agency (EMA). Product Information Beyfortus® (Nirsevimab). https://www.ema.europa.eu/en/documents/product-information/beyfortus-epar-product-information_en.pdf
8. European Medicines Agency (EMA). Product Information Synagis® (Palivizumab). https://www.ema.europa.eu/en/documents/product-information/synagis-epar-product-information_en.pdf
9. MULLER WJ, MADHI SA, SEOANE NUÑEZ B, BACA COTS M, BOSHEVA M, DAGAN R et al. Nirsevimab for prevention of RSV in term and late-preterm infants. N Engl J Med 2023; 388(16): 1533-1534. https://doi.org/10.1056/NEJMc2214773
10. ARES-GÓMEZ S, MALLAH N, SANTIAGO-PÉREZ MI, PARDO-SECO J, PÉREZ-MARTÍNEZ O, OTERO-BARRÓS MT et al. Effectiveness and impact of universal prophylaxis with nirsevimab in infants against hospitalisation for respiratory syncytial virus in Galicia, Spain: initial results of a population-based longitudinal study. Lancet Infect Dis 2024; 24(8): 817-828. https://doi.org/10.1016/S1473-3099(24)00215-9
11. ERNST C, BEJKO D, GAASCH L, HANNELAS E, KAHN I, PIERRON C et al. Impact of nirsevimab prophylaxis on paediatric respiratory syncytial virus (RSV)-related hospitalisations during the initial 2023/24 season in Luxembourg. Euro Surveill 2024; 29(4): 2400033. https://doi.org/10.2807/1560-7917.ES.2024.29.4.2400033
12. EZPELETA G, NAVASCUÉS A, VIGURIA N, HERRANZ-AGUIRRE M, JUAN BELLOC SE, GIMENO BALLESTER J et al. Effectiveness of nirsevimab immunoprophylaxis administered at birth to prevent infant hospitalisation for respiratory syncytial virus infection: A population-based cohort study. Vaccines (Basel) 2024; 12(4): 383. https://doi.org/10.3390/vaccines12040383
13. CONSOLATI A, FARINELLI M, SERRAVALLE P, ROLLANDIN C, APPRATO L, ESPOSITO S et al. Safety and efficacy of nirsevimab in a universal prevention program of respiratory syncytial virus bronchiolitis in newborns and infants in the first year of life in the Valle d'Aosta region, Italy, in the 2023-2024 epidemic season. Vaccines (Basel) 2024; 12(5): 549. https://doi.org/10.3390/vaccines12050549
14. Temporada de virus respiratorios 2023-2024 en Navarra. (Noviembre de 2024). Boletín de Salud Pública de Navarra Nº133. https://www.navarra.es/NR/rdonlyres/AECCD760-AB2A-4841-818A-FA53478FD6DC/491846/BOL133INT1.pdf
15. Instituto de Salud Pública y Laboral de Navarra. Informe Semanal 14/2025. https://www.navarra.es/NR/rdonlyres/3855C06B-B6A2-4E05-BA57-034AB7947A04/492621/InformeEpidemiologicoSemanal14_2025.pdf
16. DRYSDALE SB, CATHIE K, FLAMEIN F, KNUF M, COLLINS AM, HILL HC et al. Nirsevimab for Prevention of Hospitalizations Due to RSV in Infants. N Engl J Med 2023; 389(26): 2425-2435. https://doi.org/10.1056/NEJMoa2309189
17. LÓPEZ-LACORT M, MUÑOZ-QUILES C, MIRA-IGLESIAS A, LÓPEZ-LABRADOR FX, MENGUAL-CHULIÁ B, FERNÁNDEZ-GARCÍA C et al. Early estimates of nirsevimab immunoprophylaxis effectiveness against hospital admission for respiratory syncytial virus lower respiratory tract infections in infants, Spain, October 2023 to January 2024. Euro Surveill 2024; 29(6): 2400046. https://doi.org/10.2807/1560-7917.ES.2024.29.6.2400046
18. COMA E, MARTINEZ-MARCOS M, HERMOSILLA E, MENDIOROZ J, REÑÉ A, FINA F et al. Effectiveness of nirsevimab immunoprophylaxis against respiratory syncytial virus-related outcomes in hospital and primary care settings: a retrospective cohort study in infants in Catalonia (Spain). Arch Dis Child 2024; 109(9): 736-741. https://doi.org/10.1136/archdischild-2024-327153
19. ESTRELLA-PORTER P, BLANCO-CALVO C, LAMEIRAS-AZEVEDO AS, JUANEDA J, FERNÁNDEZ-MARTÍNEZ S, GÓMEZ-PAJARES F et al. Effectiveness of nirsevimab introduction against respiratory syncytial virus in the Valencian Community: A preliminary assessment. Vaccine 2024; 42(22): 126030. https://doi.org/10.1016/j.vaccine.2024.05.078
20. BARBAS DEL BUEY JF, ÍÑIGO MARTÍNEZ J, GUTIÉRREZ RODRÍGUEZ MÁ, ALONSO GARCÍA M, SÁNCHEZ-GÓMEZ A, LASHERAS CARBAJO MD et al. The effectiveness of nirsevimab in reducing the burden of disease due to respiratory syncytial virus (RSV) infection over time in the Madrid region (Spain): a prospective population-based cohort study. Front Public Health 2024; 12: 1441786. https://doi.org/10.3389/fpubh.2024.1441786
21. PAIREAU J, DURAND C, RAIMBAULT S, CAZAUBON J, MORTAMET G, VIRIOT D et al. Nirsevimab effectiveness against cases of respiratory syncytial virus bronchiolitis hospitalised in paediatric intensive care units in France, September 2023-January 2024. Influenza Other Respir Viruses 2024; 18(6): e13311. https://doi.org/10.1111/irv.13311
22. ASSAD Z, ROMAIN AS, AUPIAIS C, SHUM M, SCHRIMPF C, LORROT M et al. Nirsevimab and hospitalization for RSV bronchiolitis. N Engl J Med 2024; 391(2): 144-154. https://doi.org/10.1056/NEJMoa2314885
23. XU H, APARICIO C, WATS A, ARAUJO BL, PITZER VE, WARREN JL et al. Estimated Effectiveness of nirsevimab against respiratory syncytial virus. JAMA Netw Open 2025; 8(3): e250380. https://doi.org/10.1001/jamanetworkopen.2025.0380
24. MOLINE HL, TANNIS A, TOEPFER AP, WILLIAMS JV, BOOM JA, ENGLUND JA et al. Early estimate of nirsevimab effectiveness for prevention of respiratory syncytial virus-associated hospitalization among infants entering their first respiratory syncytial virus season - New Vaccine Surveillance Network, October 2023-February 2024. Morb Mortal Wkly Rep 2024; 73(9): 209-214. https://doi.org/10.15585/mmwr.mm7309a4
25. NÚÑEZ O, OLMEDO C, MORENO-PEREZ D, LORUSSO N, FERNÁNDEZ MARTÍNEZ S, PASTOR VILLALBA PE et al. Effectiveness of catch-up and at-birth nirsevimab immunisation against RSV hospital admission in the first year of life: a population-based case-control study, Spain, 2023/24 season. Euro Surveill 2025; 30(5): 2400596. https://doi.org/10.2807/1560-7917.ES.2025.30.5.2400596
26. GALA CL, HALL CB, SCHNABEL KC, PINCUS PH, BLOSSOM P, HILDRETH SW et al. The use of eye-nose goggles to control nosocomial respiratory syncytial virus infection. JAMA 1986; 256(19): 2706-2708.
27. JIANG ML, XU YP, WU H, ZHU RN, SUN Y, CHEN DM et al. Changes in endemic patterns of respiratory syncytial virus infection in pediatric patients under the pressure of nonpharmaceutical interventions for COVID-19 in Beijing, China. J Med Virol 2023; 95(1): e28411. https://doi.org/10.1002/jmv.28411
28. PASTOR-BARRIUSO R, NÚÑEZ O, MONGE S, the Nirsevimab Effectiveness Study Collaborators. Infants needed to immunise with nirsevimab to prevent one RSV hospitalisation, Spain, 2023/24 season. Euro Surveill 2025; 30(6): 2500040. https://doi.org/10.2807/1560-7917.ES.2025.30.6.2500040
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Anales del Sistema Sanitario de Navarra

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
La revista Anales del Sistema Sanitario de Navarra es publicada por el Departamento de Salud del Gobierno de Navarra (España), quien conserva los derechos patrimoniales (copyright ) sobre el artículo publicado y favorece y permite la difusión del mismo bajo licencia Creative Commons Reconocimiento-CompartirIgual 4.0 Internacional (CC BY-SA 4.0). Esta licencia permite copiar, usar, difundir, transmitir y exponer públicamente el artículo, siempre que siempre que se cite la autoría y la publicación inicial en Anales del Sistema Sanitario de Navarra, y se distinga la existencia de esta licencia de uso.
Funding data
-
Instituto de Salud Carlos III
Grant numbers FI24/00091, CP22/00016, PI20/01323, PI23/01519, INT24/00070 -
Centro Europeo para la Prevención y Control de Enfermedades
Grant numbers proyecto VEBIS







